
On March 25, 2026, Mabwell (Shanghai) Bioscience Co., Ltd., a Shanghai-based A-share listed company, submitted its prospectus to the Hong Kong Stock Exchange, planning to list on the Main Board of Hong Kong. This marks another application following the expiration of two previous filings on January 6, 2025, and August 29, 2025.
Maiwei Biotech previously received the filing approval notice from the China Securities Regulatory Commission for its overseas issuance and listing, allowing it to issue no more than 62.6646 million ordinary shares for overseas listing on the Hong Kong Stock Exchange.
Maiwei Biotech (688062.SH), listed on the A-share market on January 18, 2022, had a total market capitalization of approximately RMB 13.1 billion as of the close of trading on March 25, 2026.

 Link to Maiwei Biotech's prospectus:
Link to Maiwei Biotech's prospectus:
https://www1.hkexnews.hk/app/sehk/2026/108329/documents/sehk26032501611_c.pdf
Main Business
Maiwei Biotech, established in 2017, is a renowned Chinese pharmaceutical company recognized for its innovative capabilities in drug research and development as well as its end-to-end capabilities spanning from drug discovery to commercial sales. The company primarily focuses on independently developing drugs for oncology and age-related diseases, covering areas such as oncology, immunology, ophthalmology, and orthopedics.
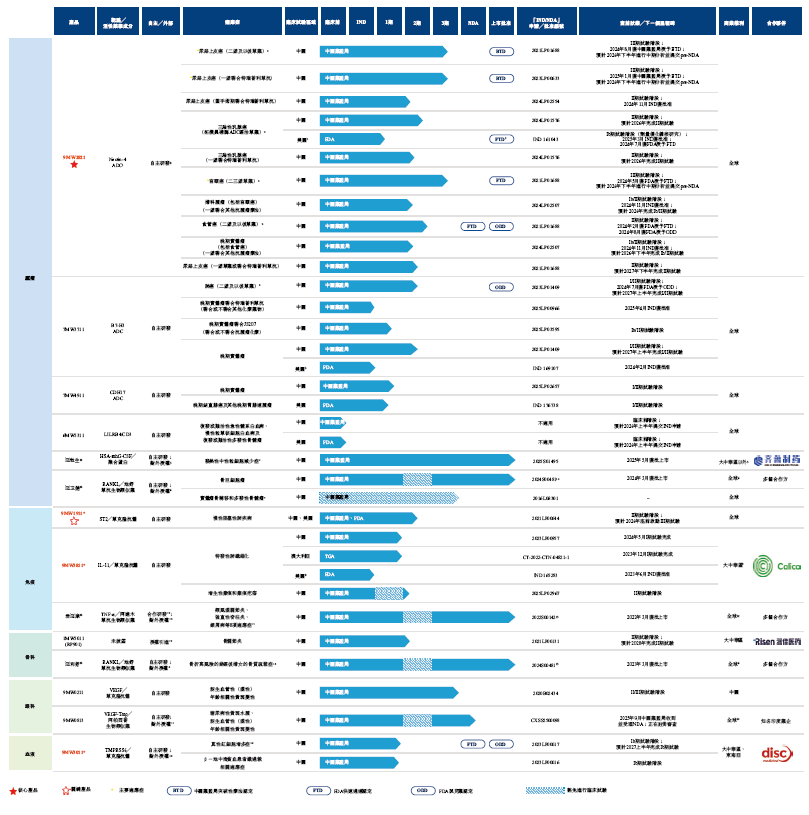
Maiwei Biotech has built a pipeline portfolio comprising more than 10 different drug assets, including:
Antibody-drug conjugates (ADCs)
Core product 9MW2821 (Bulumtatug Fuvedotin (BFv), an ADC candidate targeting Nectin-4). In terms of clinical development stages, 9MW2821 is the most advanced among all Nectin-4-targeting ADCs developed in China for treating urothelial cancer, second only to Padcev. 9MW2821 is also the world’s first Nectin-4-targeting ADC to enter the pivotal Phase III trial stage for cervical cancer. The company is conducting multiple clinical trials for 9MW2821, including (i) a Phase III trial of 9MW2821 for urothelial cancer, (ii) a Phase III trial of 9MW2821 for cervical cancer, (iii) a Phase II trial of 9MW2821 for triple-negative breast cancer, and (iv) a Phase II clinical trial of 9MW2821 for advanced esophageal cancer.
7MW3711 (targeting B7-H3 ADC), with two Phase I/II clinical trials initiated in August 2023 and September 2023 in China to evaluate the safety and efficacy of 7MW3711 as monotherapy for advanced solid tumors. Additionally, in July 2024, the FDA granted Orphan Drug Designation (ODD) for the treatment of small-cell lung cancer. In April 2025, the National Medical Products Administration (NMPA) approved the company’s IND application for a Phase Ib/II combination therapy trial of 7MW3711 combined with Toripalimab (with or without other chemotherapy drugs) for patients with advanced solid tumors. In February 2026, the company initiated a Phase Ib/II clinical trial of 7MW3711 combined with JS207 or JS207 and anti-tumor therapies for patients with advanced solid tumors.
7MW4911 (targeting CDH17 ADC). In August 2025, the FDA accepted the company’s IND application to conduct Phase I/II clinical trials for 7MW4911. In October 2025, 7MW4911 received IND approval from the National Medical Products Administration (NMPA). In November 2025, the company initiated a Phase I clinical trial for patients with advanced solid tumors. In January 2026, the company launched the clinical trial for 7MW4911 in the United States.
Other differentiated modality drugs
9MW3811 (a humanized monoclonal antibody targeting IL-11 for the treatment of fibrosis-related diseases and cancer) completed Phase I clinical trials in Australia and China in December 2023 and May 2024, respectively, involving healthy subjects. Additionally, the company obtained FDA IND approval in June 2023 for a clinical trial targeting idiopathic pulmonary fibrosis. In August 2025, the company submitted an IND application to the National Medical Products Administration (NMPA) for a clinical trial of 9MW3811 in pathological scarring (including hypertrophic scars and keloids). The NMPA granted IND approval in November 2025, making 9MW3811 the first IL-11-targeted drug to enter the clinical stage for the treatment of pathological scarring (including hypertrophic scars and keloids). In December 2025, the company initiated a clinical trial of 9MW3811 for pathological scarring (including hypertrophic scars and keloids).
The key product 9MW1911 (the first self-developed drug candidate targeting ST2 approved for clinical development in China) launched a Phase Ib/IIa trial in China in July 2023 for COPD patients, with expected completion in the first half of 2026. In July 2025, another Phase II trial for COPD patients was initiated in China, with interim analysis planned after obtaining 52-week follow-up data from at least 120 subjects, and full patient follow-up expected to be completed in the second half of 2027. The company plans to advance to a Phase III clinical trial in China in the second half of 2026 for 9MW1911 in treating COPD patients. In December 2025, the FDA granted IND approval for a Phase IIa clinical trial of 9MW1911 for COPD treatment in the United States.
9MW3011 (a recombinant humanized anti-TMPRSS6 monoclonal antibody, representing the leading global development status among TMPRSS6-targeting therapies) completed a Phase I trial in China in May 2024 involving healthy subjects. Additionally, the company plans to complete two Phase Ib clinical trials in China in the first half of 2027 for patients with polycythemia vera.
1MW5011 (RP901) (a small-molecule drug for osteoarthritis treatment) is currently recruiting patients for a Phase II clinical trial for knee osteoarthritis, with expected completion by 2028. Additionally, the company initiated a Phase Ib trial in December 2025 for patients with knee osteoarthritis.
9MW0211 (a recombinant humanized monoclonal antibody targeting VEGF-A) completed a Phase I trial for ophthalmic diseases in May 2019. The Phase I trial results demonstrated that 9MW0211 has good safety and efficacy. The company initiated a Phase II/III clinical trial in China in May 2021, completed it in December 2025, and plans to submit an NDA to the National Medical Products Administration (NMPA) at an appropriate time.
9MW0813 (a biosimilar of the recombinant human VEGF receptor-antibody fusion protein aflibercept) completed its Phase III clinical trial in December 2024, and an NDA was submitted to the National Medical Products Administration (NMPA), which was accepted in September 2025.
Marketed drugs
Four products of Maiwei Bio—Junmaikang®, Maiweijian® (oncology), Mailisu® (orthopedics), and Mailisheng® (febrile neutropenia)—have been successfully launched.
Maiwei Bio's revenue comes from pharmaceutical sales and out-licensing income.

Shareholder Structure
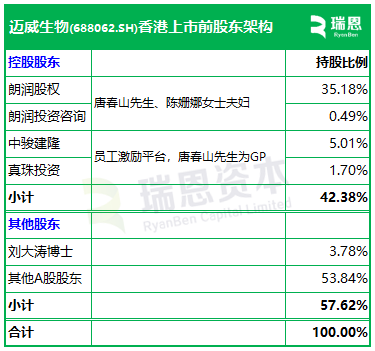
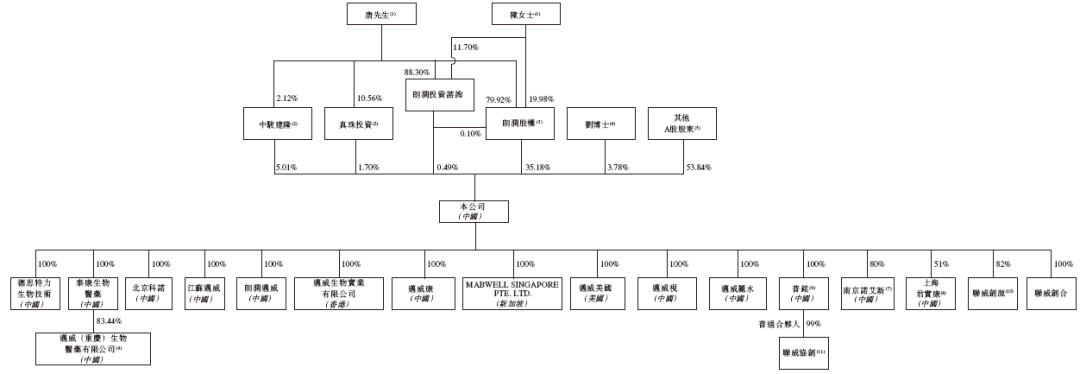
The prospectus shows that, prior to the listing, the shareholder structure of Maiwei Bio included:
Mr. Tang Chunshan and Mrs. Chen Shanna respectively held 35.18%, 0.49%, 5.01%, and 1.74% of the shares through Langrun Equity, Langrun Investment Consulting, Zhongjun Jianlong (an employee incentive platform with Mr. Tang as the GP), and Zhenzhu Investment (an employee incentive platform with Mr. Tang as the GP), collectively holding approximately 42.38%.
Dr. Liu Datao held 3.78% of the shares;
Other A-share shareholders held 53.84% of the shares.
Directors and senior management
The board of directors of Maiwei Bio consists of 10 members, including:
Five executive directors:
Mr. Tang Chunshan (controlling shareholder);
Dr. Liu Datao (chairman and general manager);
Dr. Wu Hai (vice general manager and R&D president);
Mr. Hu Huiguo (vice general manager and board secretary);
Dr. Gui Xun (employee representative director and vice general manager);
One non-executive director:
Mr. Wu Yufeng (Managing Partner of Hainan Shiyu Private Equity);
Four independent non-executive directors:
Mr. Qin Zhengyu (Chief Financial Officer and Vice General Manager of Zijiang Enterprise (600210.SH));
Dr. Xu Qing (Vice Chairman of the Chinese Association of Medical Biotechnology);
Dr. Zhao Qian (Professor at Shanghai Jiao Tong University School of Medicine);
Ms. Wang Fang (Executive Director of Hengkuan International Group).
In addition to the executive directors, senior executives include:
Dr. Wang Shuhai (Vice General Manager);
Mr. Li Han (Vice General Manager);
Mr. Ni Hua (Vice General Manager);
Ms. Chen Xi (Vice General Manager);
Mr. Hua Jun (Vice General Manager and Financial Controller).
Company performance
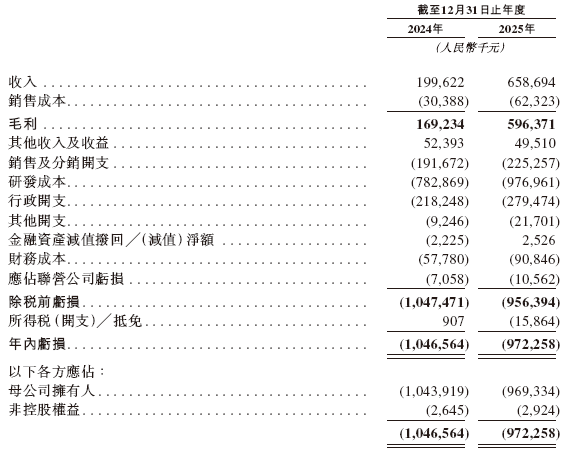
The prospectus shows that in the past 2024 and 2025, Maiwei Bio's operating revenues were RMB 2.00 billion and RMB 6.59 billion respectively, corresponding research and development expenditures were RMB 7.83 billion and RMB 9.77 billion respectively, and corresponding net losses were RMB 1.047 billion and RMB 972 million respectively.
Intermediary team
The intermediary team for Maiwei Bio's IPO this time mainly includes:
CITIC Securities and Haitong International are its joint sponsors;
EY as its auditor;
Jingtian & Gongcheng served as its company's legal counsel in China.
Davis Polk & Wardwell as its legal counsel in Hong Kong and the United States;
King & Wood Mallesons as its China legal counsel for the underwriter;
Herbert Smith Freehills serves as its legal counsel for securities in Hong Kong and the United States;
Sunbase Capital as its compliance advisor;
Frost & Sullivan serves as its industry consultant.
