Securities firms believe that the innovative drug sector is in a phase of 'performance realization, valuation recovery, and conference-driven catalysts.' Several biopharmaceutical companies have achieved historical turnarounds, with fundamental improvements. In the first quarter of this year, the total value of overseas licensing for China's innovative drugs reached nearly USD 570 billion, surpassing the full-year level of 2024. The upcoming AACR and ASCO annual meetings are expected to provide catalysts through the release of dense clinical data.
Signs of an explosive breakout in the innovative drug sector have emerged since last week. Hong Kong-listed stocks $CHINARES PHARMA (03320.HK)$ 、 $REMEGEN (09995.HK)$ 、 $INNOCARE (09969.HK)$ have surged over 20% cumulatively in the past five trading sessions, $SIMCERE PHARMA (02096.HK)$ 、 $HANSOH PHARMA (03692.HK)$ 、 $ASCLETIS-B (01672.HK)$ while others have risen by more than 17% cumulatively.
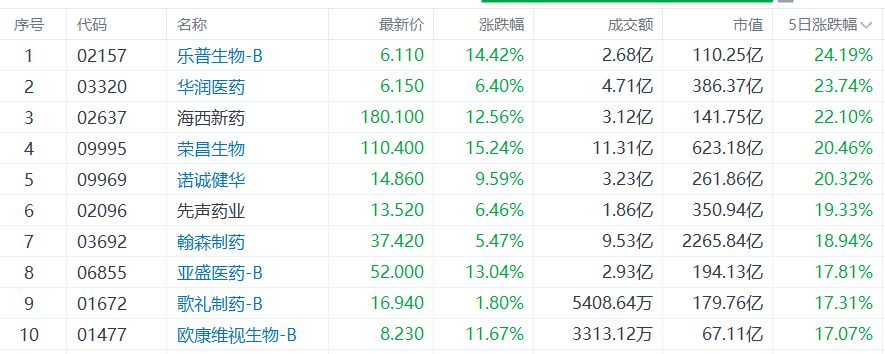
Several brokerage firms pointed out that the innovative drug sector is currently in a favorable phase characterized by 'earnings realization, valuation recovery, and conference catalysts'.
 Specifically, leading pharmaceutical companies delivered impressive 2025 annual reports. Companies such as Hengrui Pharma and BeiGene have seen a continuous increase in the proportion of revenue from innovative drugs. Meanwhile, several biopharmaceutical enterprises, including InnoCare Pharma and RemeGen, achieved historic turnarounds. Additionally, the upcoming AACR Annual Meeting in April and ASCO Annual Meeting in May-June are expected to provide ongoing momentum for valuation recovery through intensive clinical data disclosures.
Specifically, leading pharmaceutical companies delivered impressive 2025 annual reports. Companies such as Hengrui Pharma and BeiGene have seen a continuous increase in the proportion of revenue from innovative drugs. Meanwhile, several biopharmaceutical enterprises, including InnoCare Pharma and RemeGen, achieved historic turnarounds. Additionally, the upcoming AACR Annual Meeting in April and ASCO Annual Meeting in May-June are expected to provide ongoing momentum for valuation recovery through intensive clinical data disclosures.
Innovative drug leaders experience qualitative changes in revenue structure, with 'turnaround' emerging as a key term.
The 2025 annual report indicates that China's innovative drug industry is undergoing structural changes.
$HENGRUI PHARMA (01276.HK)$ As the leading company in China’s innovative drug industry, it achieved revenue of RMB 31.629 billion in 2025, representing a year-on-year increase of 13.02%; net profit attributable to shareholders reached RMB 7.711 billion, marking a year-on-year growth of 21.69%.
The most remarkable indicator in the financial report was the revenue from innovative drug sales, which reached 16.342 billion yuan, increasing by 26.09% year-on-year. The proportion of innovative drug revenue in total pharmaceutical sales rose to 58.34%, making innovative drugs the core growth driver for the company. Moreover, the external licensing business of innovative drugs contributed revenue of 3.392 billion yuan, forming the second growth curve following self-operated sales.
$FOSUN PHARMA (02196.HK)$ In 2025, revenue from innovative drugs reached RMB 9.893 billion, increasing by 29.59% year-on-year, accounting for 23.75% of total revenue, up 5.16 percentage points year-on-year.
More notably, a group of biopharmaceutical companies turned profitable in 2025. $BEONE MEDICINES (06160.HK)$ According to its annual report, the company generated revenue of USD 5.343 billion (approximately RMB 38.7 billion) in 2025, representing a year-on-year increase of 40.2%; net profit was USD 287 million, compared to a net loss of USD 645 million in the same period last year, marking a historic turning point toward profitability.
Shenwan Hongyuan Securities noted in its pharmaceuticals industry weekly report released on March 29 that, as of the same date, 71 pharmaceutical listed companies had published their 2025 annual reports. Among them, 32 companies achieved positive growth in net profit attributable to parent company shareholders, with 10 companies reporting a growth rate of 40% or higher, demonstrating both overall profitability recovery and structural differentiation within the sector.
Surge in BD Transactions: First-quarter outbound licensing value surpasses total for 2024
Another critical logic line for the innovative drug industry is the continued vibrancy of outbound licensing (BD) transactions.
According to statistics from the NextPharma database of PharmaCube, as of March 25, 2026, there have been 49 outbound BD events in China's innovative drug sector, with upfront payments exceeding $3.3 billion and total transaction value nearing $57 billion—indicating that the scale of transactions in just the first quarter has already surpassed the total for the entire year of 2024 and is approaching half of the total for 2025.
This data suggests that the R&D capabilities of China’s innovative pharmaceutical companies are gaining global recognition, with overseas licensing emerging as another significant revenue stream following self-commercialization. The contribution of outbound licensing to Hengrui Pharma’s 2025 financial report, amounting to RMB 3.392 billion in revenue, epitomizes this trend.
Valuation recovery bolstered by policy support creates an opportune time for innovative drug investment
Multiple brokerages in their research reports issued this week generally believe that the innovative drug sector presents an excellent opportunity for strategic allocation.
Huaxin Securities pointed out that the development model of innovative drugs has shifted from reliance solely on financing to a comprehensive approach combining product commercialization, R&D-driven outbound licensing, and financing, signaling an ideal time for allocation. Meanwhile, after undergoing over half a year of adjustments since the end of Q3 last year, valuation risks in the innovative drug sector have been substantially mitigated, and short-term sentiment pressures do not alter the fundamental long-term growth trajectory of the industry.
On the policy front, the 2026 'Two Sessions' positioned the pharmaceuticals and biotechnology sector as an emerging pillar industry alongside integrated circuits and aerospace, proposing to build global competitiveness. This policy orientation lays a solid foundation for the long-term development of the innovative drug industry.
With the upcoming AACR and ASCO conferences, clinical data releases are expected to act as catalysts for valuation recovery.
The academic conferences over the next two months are expected to serve as short-term catalysts for the innovative drug sector.
From April 17 to 22, the American Association for Cancer Research (AACR) Annual Meeting will take place. In 2025, a total of 126 Chinese pharmaceutical companies will present at AACR, showcasing nearly 300 new drug research findings, including over 80 ADC studies and nearly 40 bispecific antibody studies.
From May 29 to June 2, the American Society of Clinical Oncology (ASCO) Annual Meeting will also be held. In 2025, more than 70 oral presentations from China have been selected, with 11 included in the highly prestigious Late-Breaking Abstracts (LBA).
GF Securities believes that $AKESO (09926.HK)$ PD-1/VEGF bispecific antibodies, $SKB BIO-B (06990.HK)$ TROP2-ADC, $DUALITYBIO-B (09606.HK)$ B7H3-ADC, $CSPC PHARMA (01093.HK)$ EGFR-ADC, $INNOCARE (09969.HK)$ Key clinical data for major products such as BCL-2 inhibitors disclosed at the aforementioned conferences are expected to further serve as catalysts for valuation recovery.
Editor/KOKO
